Google shows the future of the bathroom: toilet can also help you detect the body
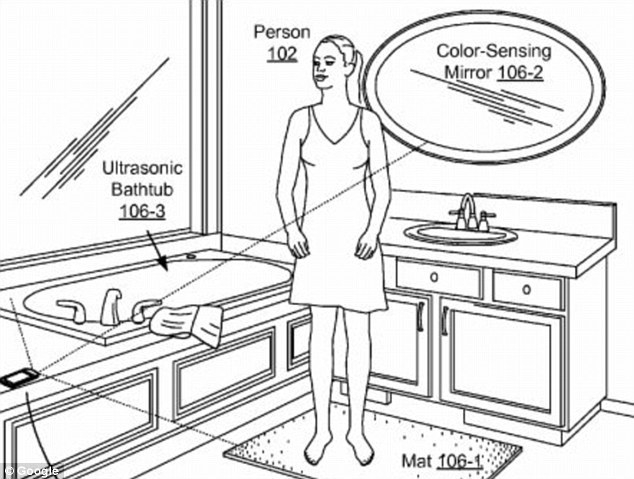
According to the British "Daily Mail" report, Google recently announced a patent that describes the future of smart bathroom. The bathroom is equipped with multiple non-invasive health monitoring devices, including ultrasonic baths and pressure-sensing toilets, which can fully monitor the user's cardiovascular health. According to the patent, this technology can ascertain the “functional status and trend of the human physiological system†so that it can warn people before the disease is formed.
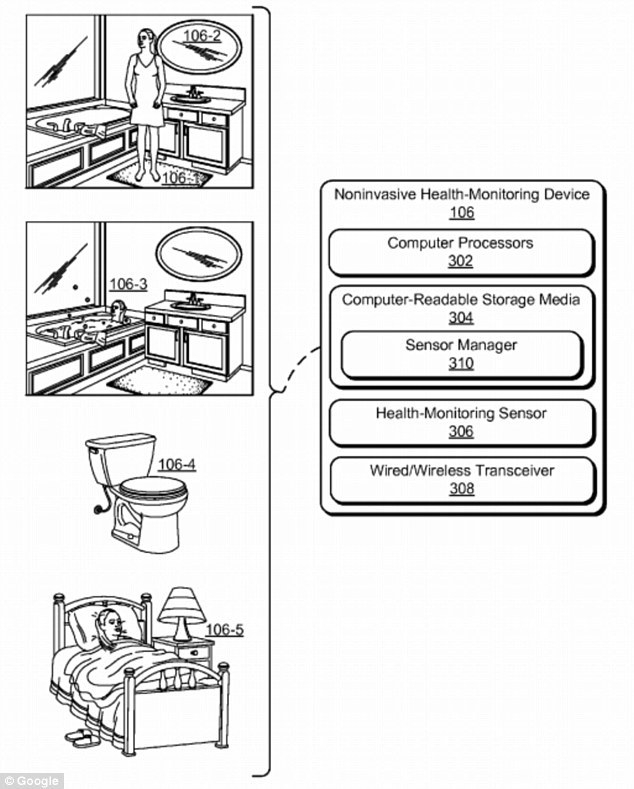
The bathroom is equipped with a series of sensors and cameras for instruments such as pressure sensing carpets, color-sensing mirrors and ultrasonic baths:
An electric force sensing carpet can sense the pulse wave velocity of human blood.
The pressure toilet seat can not only measure blood pressure, but also analyze the body's excretion.
The color-sensing mirror in the bathroom can sense the color of the skin through a specific waveform, and record the shape of the body or the internal organs and changes in color.
Ultrasonic baths can generate high-frequency sound waves and collect sound wave reflections to probe blood flow and tissue movement in the human body. At the same time, it can also perform three-dimensional measurements on the body's internal structure.
The radar device is used to receive the radiation released by the tissues in the human body and to measure the human skin temperature, heart rate, and bone movement data.
The advantage of bioelectronics technology is that it can detect the health status of patients in real time and help pharmaceutical companies to develop disease interventions. At present, Google's patent is only for cardiovascular diseases, and officials said that they will be applied to the human nerve, endocrine, muscle, bone, or epidermis system in the future. Last week, Google and pharmaceutical company GlaxoSmithKline conducted in-depth cooperation to jointly develop bio-electronics products.
Of course, they still face many challenges. First of all, for Google, which has always been not good at hardware development, it will take time to convert this patent into a product. At the same time, it is not easy for technology to obtain approval from regulators including the US Food and Drug Administration (FDA). Even if the product is molded and meets the above conditions, the safety and effectiveness of the medical product must be observed and tried in the clinic for a long time before it can be used on a large scale.

Working Cannula,Percutaneous Discectomy Working Cannula,Following Surgical Operation Procedures,Surrounding Guide Wire
Dragon Crown Medical Co., Ltd. , https://www.dragoncrownmed.com